Automotive Battery Explosions
(Published in Property Casualty 360, January 23, 2013)
By
Charles C. Roberts, Jr., Ph.D., P.E.
As shown in Figure 1, the typical automotive battery, of lead/acid construction, is an electrochemical container that produces voltage, which causes electrical current to flow to various components in an automotive vehicle. An outer polymer case (high density polypropylene) acts as a container for an electrolyte (sulfuric acid), six cells and lead plates. Each cell delivers 2.1 volts with a total voltage of 12.6 volts, at full charge. Vents are installed at the top of the battery to vent gasses formed during the normal charging cycles.
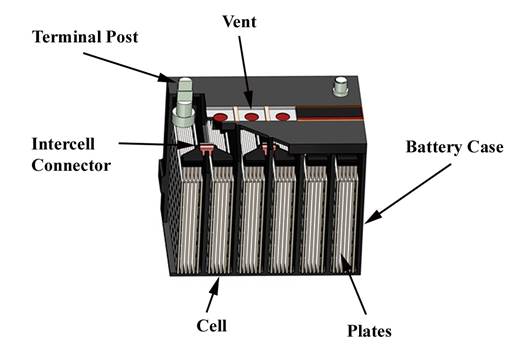
Figure 1
During the charging cycle, hydrogen gas is generated and accumulates in the head space above the electrolyte level, prior to venting. Hydrogen gas has a wide range of explosive limits in air, ranging from 4 to 72% hydrogen in air and is easily ignited by a flame or spark. If the hydrogen is ignited inside the battery, it typically blows off the top of the battery case, showering sulfuric acid in the immediate vicinity along with fragments of the battery case. The explosive energy generation is so rapid that the vents cannot relieve the pressure in time to prevent an explosion.
Figure 2 is a view of a two year old battery that exploded, causing personal injury from acid burns. This occurred when a standby generator was starting during its normal maintenance test cycle. The top of the battery was blown away, suggesting that hydrogen was ignited inside the battery. Figure 3 is a view of a battery fragment that was found imbedded in the ceiling of the building that enclosed the generator.

Figure 2

Figure 3

Figure 4
Inspection of the battery shown in Figure 2 revealed that one of the intercell connectors (Figure 4) was loose and corroded. A loose connection inside a battery can result in an electrical arc jumping across the gap, igniting the hydrogen. In this case, the loose connection was determined to be a result of a manufacturing defect.
Other internal explosive ignition conditions may exist that are not related to a manufacturing defect:
· A conductive bridge may be formed between two plates as a result of low electrolyte levels. When a high current demand is placed on the battery, it can arc, igniting hydrogen gas and initiating an explosion. This is a result of improper battery maintenance where the electrolyte level should be monitored periodically and lost electrolyte replaced. Maintenance free batteries have a hydrometer that measures the specific gravity of the electrolyte (an indicator of the concentration of the electrolyte and hence, the electrolyte level) and indicates whether or not a battery should be replaced.
· External ignition sources often manifest themselves in the form of loose battery cable connections or a poor connection with battery charger clamps that generate an electrical arc. Jump starting vehicles with dead batteries can result in electrical arcs at the dead battery terminals if the last set of clamps is attached to the dead battery. Recommended procedure is to attach the jumper clamps to the dead battery first and then to the live battery.
· Battery explosions have occurred as a result of tools being placed between the battery terminals. Some individuals test a battery by placing a screw driver across the terminals to see if an arc jumps, revealing whether the battery is supplying electrical energy or not. This can result in a battery explosion since the current through the screw driver is not regulated, can be very high and generate an electrical arc, causing an internal or external explosion.
· Corrosion, which can cause electrical resistance and a possible arc ignition source, may develop at battery terminals as shown in Figure 5, which also illustrates a loose or improperly secured battery clamp.

Figure 5
As in most investigations, retention of the evidence is desirable if subrogation is anticipated. In battery explosion losses, fragments may be found scattered throughout the scene and imbedded in the building structure. Collecting these pieces helps the technical analyst determine the cause of the explosion. It is encouraged to have the battery analyzed in a timely manner to reduce the effect of corrosion, which can obscure evidence.